 The presence of the enzyme family called carbonic anhydrases in the red blood cells will result in an increase in reaction rate. The hydration reactions undergone by carbon dioxide are very slow, especially in the absence of a suitable catalyst. The bicarbonate ion or molecule is an intermediate for the carbon dioxide out of the human body through the respiratory gas exchange process. It can be orally consumed to induce vomiting whenever required (such as, in the case of a drug overdose). Solutions that contain this compound are much more effective in contact lens cleaning. Ringworm and other dermatitides are treated through the carbonic acid application over the affected area. H 2 CO 3 protonates many bases that contain nitrogen in blood serum It helps in carbon dioxide transportation out of the body. H 2 CO 3 can be used in the precipitation of various ammonium salts like ammonium persulfate. The preparation of sparkling wine, carbonated water, and other aerated drinks involves carbonic acid use. A few of the carbonic acid uses are listed below: H 2 CO 3 is an essential compound with a wide range of applications. It should also note that carbonic acid can be obtained as the burning of fossil fuels at an industrial scale or a by-product of industrial fermentation processes. In the presence of water, it undergoes partial dissociation to yield HCO 3 – (bicarbonate) and H + ions.Ĭarbonic acid is a diprotic acid, and hence, it can form two types of salts: carbonates and bicarbonates.Īdding a small quantity of base to H 2 CO 3 yields bicarbonate salts, whereas the addition of a base in excess amounts yields carbonate salts. H 2 CO 3 is unstable in nature, and it is a weak acid. The conjugate base corresponding to the carbonic acid is bicarbonate. But, it has been reported that NASA scientists have prepared solid H 2 CO 3 samples. Generally, this compound exists as a solution. It has a density of 1.668 grams per cubic centimetre in a standard state. The H 2 CO 3 compound holds a pKa value of 3.6. The carbonic acid molar mass can be given as 62.024 grams per mole. However, in the chemical equilibrium described above, only a small amount of carbon dioxide is converted into carbonic acid.Ī few major physical and chemical properties of carbonic acid are listed in this subsection: When CO 2 is dissolved in water, it participates in the chemical equilibrium as given below. 
Carbonic acid is both unstable, weak and dissociates quickly into the bicarbonate ions (HCO 3 – ) and hydrogen ions (H + ). The oxygen atoms that participate in a single bond with the carbon each contain one hydrogen atom attached to them.Ĭarbonic acid, formed by the hydrolysis and dissolution of CO 2 in water, is the primary natural leaching agent in various temperate ecosystems. The carbonic acid or H 2 CO 3 structure can be illustrated below.įrom the illustration above, we can understand that the carbonic acid structure consists of one carbon-oxygen double bond and two carbon-oxygen single bonds. 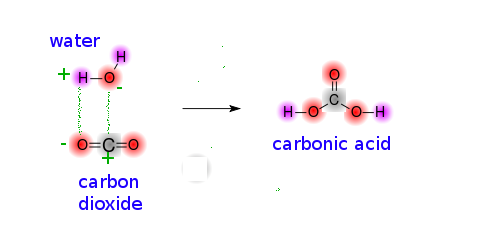
This is the primary reason for various features of limestone, including stalactites and stalagmites. Note: H 2 CO 3 is capable of dissolving limestone, which leads to the calcium bicarbonate (Ca(HCO 3 ) 2 formation. It forms bicarbonate and carbonate salts, and it is a weak acid. The carbonic acid's chemical formula can also be written as OC(OH) 2 because, in this compound, there is one carbon-oxygen double bond.Ĭarbonic acid can often be described as a respiratory acid due to the reason it is the only acid exhaled by the human lungs in the gaseous state. The carbon dioxide solutions in water comprise small amounts of this compound. 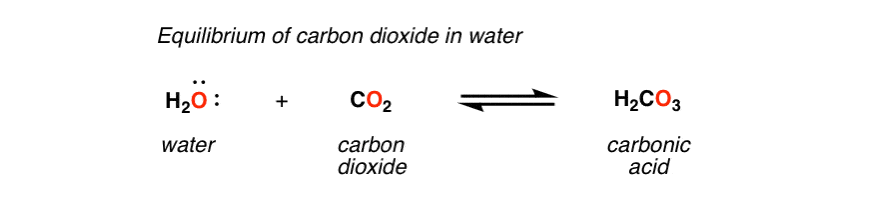
Carbonic acid can be defined as a carbon-containing compound having the chemical formula H 2 CO 3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
